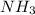Chemistry, 20.10.2019 04:30 hannaharsena
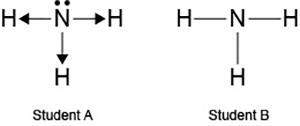
Two students made the lewis dot diagrams of nh3. the diagrams are as shown.
which student drew the correct lewis dot diagram?
only student a
only student b
both student a and student b
neither student a nor student b

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3


You know the right answer?
Two students made the lewis dot diagrams of nh3. the diagrams are as shown.
which student...
which student...
Questions



Biology, 14.10.2019 12:30










English, 14.10.2019 12:30


Social Studies, 14.10.2019 12:30

Biology, 14.10.2019 12:30




Mathematics, 14.10.2019 12:30