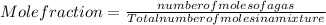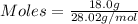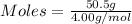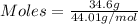Chemistry, 22.10.2019 02:00 jstringe424
Asample contains 18.0 g n2 (mw = 28.02 g/mol), 50.5 g he (mw = 4.00 g/mol), and 34.6 g co2 (mw = 44.01 g/mol). calculate the mole fraction of carbon dioxide in the sample.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:20
Determine which intermolecular forces are the dominant (strongest) forces for a pure sample of each of the following molecules by placing the molecules into the correct bins. drag the appropriate molecular formula to their respective bins.
Answers: 3

Chemistry, 21.06.2019 23:00
When determining the shape of a molecule, it is important to draw a lewis dot structure first in order to see the total number a. electrons within the moleculeb. bonding and unshared pairs around central atomc. unshared pair within the molecule( i really need it )
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 23:30
To find the work done, the force exerted and distance moved are multiplied. a couch is moved twice before you are happy with its placement. the same force was used to move the couch both times. if more work is done the first time it is moved, what do you know about the distance it was moved? a) when more work was done, the couch was moved the same distance. b) when more work was done, the couch was moved less. c) when more work was done, the couch was moved further. d) when more work was done, the couch wasn't moved at all.
Answers: 1
You know the right answer?
Asample contains 18.0 g n2 (mw = 28.02 g/mol), 50.5 g he (mw = 4.00 g/mol), and 34.6 g co2 (mw = 44....
Questions

Mathematics, 15.05.2021 03:10




Business, 15.05.2021 03:10


Mathematics, 15.05.2021 03:10




Mathematics, 15.05.2021 03:10



Mathematics, 15.05.2021 03:10


Biology, 15.05.2021 03:10

Chemistry, 15.05.2021 03:10



Mathematics, 15.05.2021 03:10