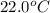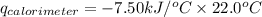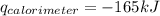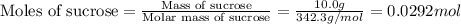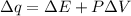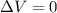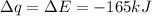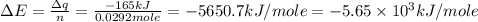Consider the reaction c12h22o11(s)+12o2(g)→12co2(g)+11h2o (l) in which 10.0 g of sucrose, c12h22o11, was burned in a bomb calorimeter with a heat capacity of 7.50 kj/∘c. the temperature increase inside the calorimeter was found to be 22.0 ∘c. calculate the change in internal energy, δe, for this reaction per mole of sucrose. express the change in internal energy in kilojoules per mole to three significant figures.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 23.06.2019 03:00
Which of the following is a chemical property of water at 4 c
Answers: 2

Chemistry, 23.06.2019 13:30
Malik formed a hypothesis that an increase in atmospheric oxygen levels by 10% would cause red-legged grasshoppers to grow larger than normal. suppose that malik performs an experiment to test his hypothesis. which of these actions would represent a scientific mistake in his experiment? a. he experiments on live grasshoppers instead of preserved ones. b. he focuses on red-legged grasshoppers instead of all kinds of grasshoppers. c. he varies the nitrogen and carbon dioxide levels in the air from one trial to the next. d. he conducts the experiment in a controlled lab setting with a lab partner. e. he measures the mass and length of his specimens at the start of each trial.
Answers: 1

Chemistry, 23.06.2019 18:00
An engineer designing a new type of engine needs a liquid that can be heated and cooled quickly with as little exchange of energy as possible. which property should the engineer primarily look for in the liquid? a. low thermal conductivity b. low specific heat capacity c. high reactivity d. high internal energy e. high density
Answers: 1
You know the right answer?
Consider the reaction c12h22o11(s)+12o2(g)→12co2(g)+11h2o (l) in which 10.0 g of sucrose, c12h22o11,...
Questions

Mathematics, 15.01.2021 21:20


English, 15.01.2021 21:20


Mathematics, 15.01.2021 21:20

History, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20



Social Studies, 15.01.2021 21:20


Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20

Mathematics, 15.01.2021 21:20


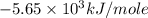
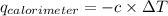
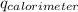 = heat released by calorimeter = ?
= heat released by calorimeter = ?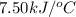
 = change in temperature of calorimeter =
= change in temperature of calorimeter =