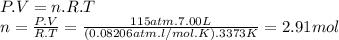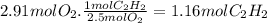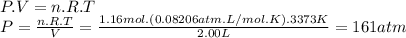Chemistry, 22.10.2019 23:00 kihgff5711
Magine that you have a 7.00 l gas tank and a 2.00 l gas tank. you need to fill one tank with oxygen and the other with acetylene to use in conjunction with your welding torch. if you fill the larger tank with oxygen to a pressure of 115 atm , to what pressure should you fill the acetylene tank to ensure that you run out of each gas at the same time? assume ideal behavior for all gases.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
What is driving behind plate tectonics (plate movment)? a) gravity only b) inertia c) convection and gravity d) the sun theres no option for science so i picked chemistry. plz
Answers: 2



Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
Magine that you have a 7.00 l gas tank and a 2.00 l gas tank. you need to fill one tank with oxygen...
Questions

Health, 18.09.2019 06:00



Biology, 18.09.2019 06:00


Social Studies, 18.09.2019 06:00

Biology, 18.09.2019 06:00


History, 18.09.2019 06:00

History, 18.09.2019 06:00



History, 18.09.2019 06:00


Mathematics, 18.09.2019 06:00


Mathematics, 18.09.2019 06:00


Social Studies, 18.09.2019 06:00

Chemistry, 18.09.2019 06:00