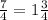Chemistry, 22.10.2019 23:00 kaitlynmorgan43
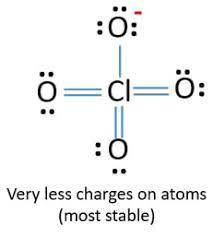
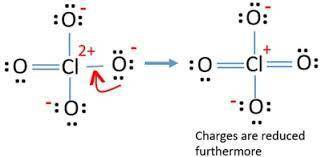
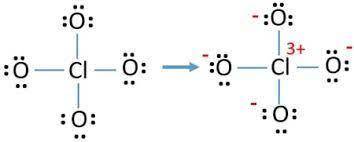
Lewis structures for the perchlorate ion (clo4−) can be drawn with all single bonds or with one, two, or three double bonds. draw each of these possible resonance forms, including any nonbonding electrons. include the values of any nonzero formal charges. use formal charges to determine the most important resonance structure and calculate its average bond order. slader

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
Lewis structures for the perchlorate ion (clo4−) can be drawn with all single bonds or with one, two...
Questions



Mathematics, 20.03.2021 01:00


Mathematics, 20.03.2021 01:00


Mathematics, 20.03.2021 01:00

Mathematics, 20.03.2021 01:00

Mathematics, 20.03.2021 01:00






History, 20.03.2021 01:00

Mathematics, 20.03.2021 01:00

Mathematics, 20.03.2021 01:00


Chemistry, 20.03.2021 01:00

 or
or  .
.