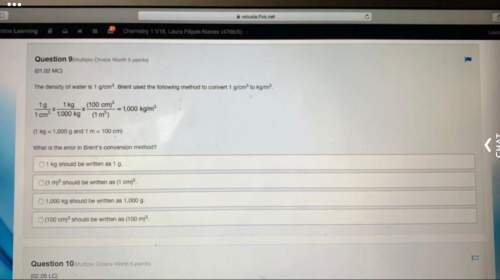
At 100 ∘c the equilibrium constant for the reaction cocl2(g)←−→co(g)+cl2(g) has the value kc=2.19×10−10. are the following mixtures of cocl2, co, and cl2 at 100 ∘c at equilibrium? if not, indicate the direction that the reaction must proceed to achieve equilibrium. part a [cocl2]=2.00×10−3m, [co]=3.3×10−6m, [cl2]=6.62×10−6m , , the reaction is at equilibrium. the reaction will proceed right to attain equilibrium. the reaction will proceed left to attain equilibrium.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a reaction has g of -136kj at 110°c, will it be spontaneous at this temperature (110°c)? yes or no
Answers: 2

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

You know the right answer?
At 100 ∘c the equilibrium constant for the reaction cocl2(g)←−→co(g)+cl2(g) has the value kc=2.19×10...
Questions

Computers and Technology, 02.12.2021 20:30

Computers and Technology, 02.12.2021 20:30

Computers and Technology, 02.12.2021 20:30

Geography, 02.12.2021 20:30


Mathematics, 02.12.2021 20:30

Computers and Technology, 02.12.2021 20:30


Mathematics, 02.12.2021 20:30

History, 02.12.2021 20:30

Medicine, 02.12.2021 20:30


Mathematics, 02.12.2021 20:30


History, 02.12.2021 20:30

Social Studies, 02.12.2021 20:30


Social Studies, 02.12.2021 20:30

Health, 02.12.2021 20:30

![Q=\frac{[CO].[Cl_{2}]}{[COCl_{2}]} =\frac{3.3 \times 10^{-6} \times 6.62 \times 10^{-6} }{2.00 \times 10^{-3}} =1.09 \times 10^{-8}](/tpl/images/0342/7533/b0aa3.png)