
Chemistry, 23.10.2019 23:00 robbinsjeffrey271
Asample of nitrosyl bromide (nobr) decomposes according to the equation 2nobr(g)⇌2no(g)+br2(g) an equilibrium mixture in a 5.00-l vessel at 100 ∘c contains 3.29 g of nobr, 3.04 g of no, and 8.10 g of br2. part apart complete calculate kc. express your answer using three significant figures. 0.116 previous answers correct part bpart complete what is the total pressure exerted by the mixture of gases? express your answer to three significant figures and include the appropriate units. 1.11 atm previous answers correct part c what was the mass of the original sample of nobr?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

You know the right answer?
Asample of nitrosyl bromide (nobr) decomposes according to the equation 2nobr(g)⇌2no(g)+br2(g) an eq...
Questions

History, 19.03.2021 19:30



Mathematics, 19.03.2021 19:30

Mathematics, 19.03.2021 19:30







Mathematics, 19.03.2021 19:30




Chemistry, 19.03.2021 19:30

History, 19.03.2021 19:30

Mathematics, 19.03.2021 19:30

History, 19.03.2021 19:30

![kc = \frac{[Br_{2}][NO]^2{[NOBr]^2}](/tpl/images/0343/4270/3b92b.png) (1)
(1)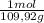 = 0,0299 moles/ 5,00L = 5,986x10⁻³M
= 0,0299 moles/ 5,00L = 5,986x10⁻³M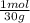 = 0,101 moles/ 5,00L = 0,0203M
= 0,101 moles/ 5,00L = 0,0203M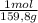 = 0,0507 moles/ 5,00L = 0,0101M
= 0,0507 moles/ 5,00L = 0,0101M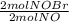 = 0,101 mol NOBr
= 0,101 mol NOBr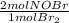 = 0,101 mol NOBr
= 0,101 mol NOBr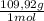 = 25,5 g of NOBr
= 25,5 g of NOBr


