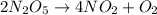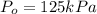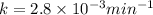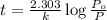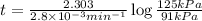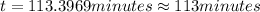The decomposition of dinitrogen pentoxide, n2o5, to no2 and o2 is a first-order reaction. at 60°c, the rate constant is 2.8 × 10-3min-1. if a rigid vessel initially contains only n2o5 at a pressure of 125 kpa, how long will it take for the total pressure to reach 176 kpa? options (pick 1)a)113 minb)129 minc)42 mind)182 mine)62 minf)83 min

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3



Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
You know the right answer?
The decomposition of dinitrogen pentoxide, n2o5, to no2 and o2 is a first-order reaction. at 60°c, t...
Questions




Social Studies, 30.06.2021 04:50





Computers and Technology, 30.06.2021 04:50

Chemistry, 30.06.2021 04:50

Mathematics, 30.06.2021 04:50

Mathematics, 30.06.2021 04:50

Arts, 30.06.2021 04:50

Business, 30.06.2021 04:50





Mathematics, 30.06.2021 04:50

Chemistry, 30.06.2021 04:50