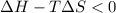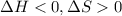Chemistry, 27.10.2019 03:43 Ezekielcassese
Favorable (exergonic) chemical reactions:
a) are generally explosive
b) absorb energy from the surroundings
c) occur only in the gaseous phase
d) usually give off heat energy

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3

Chemistry, 23.06.2019 08:00
Which term means two or more atoms that share electrons in a chemical bond? a. hydrogen bond b. moleculec. ionic bondd. element amd you
Answers: 3
You know the right answer?
Favorable (exergonic) chemical reactions:
a) are generally explosive
b) absorb e...
a) are generally explosive
b) absorb e...
Questions



Health, 01.04.2021 19:00


English, 01.04.2021 19:00



History, 01.04.2021 19:00


Mathematics, 01.04.2021 19:00








Mathematics, 01.04.2021 19:00

Health, 01.04.2021 19:00

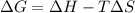
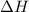 is the enthalpy change;
is the enthalpy change;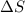 is the entropy change;
is the entropy change;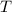 is the absolute temperature.
is the absolute temperature.