Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1
You know the right answer?
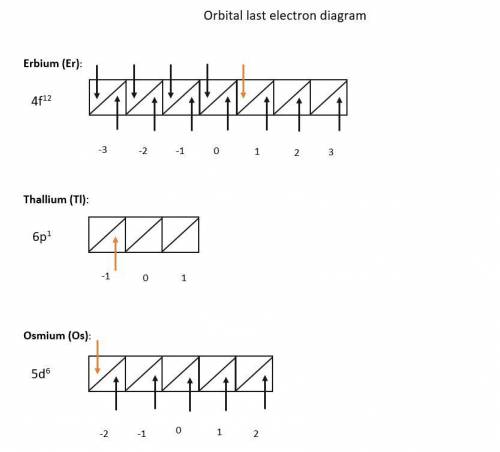
The quantum numbers for the last electron placed in three elements are listed below. which of these...
Questions


Biology, 17.02.2020 07:13


Mathematics, 17.02.2020 07:13

Mathematics, 17.02.2020 07:13

World Languages, 17.02.2020 07:13



Mathematics, 17.02.2020 07:14


History, 17.02.2020 07:16

World Languages, 17.02.2020 07:17





Biology, 17.02.2020 07:23

History, 17.02.2020 07:26

Mathematics, 17.02.2020 07:26