
If the aluminum block is initially at 25 ∘c, what is the final temperature of the block after the evaporation of the alcohol? assume that the heat required for the vaporization of the alcohol comes only from the aluminum block and that the alcohol vaporizes at 25 ∘c. the heat of vaporization of the alcohol at 25 ∘c is 45.4 kj/mol, the specific heat of aluminum is 0.903 j/g⋅∘c

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Noble gases are the most reactive elements on the periodic table. a. true b. false
Answers: 2

Chemistry, 21.06.2019 18:10
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1
You know the right answer?
If the aluminum block is initially at 25 ∘c, what is the final temperature of the block after the ev...
Questions


Mathematics, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Social Studies, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57







Geography, 11.06.2020 02:57


History, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Mathematics, 11.06.2020 02:57

Chemistry, 11.06.2020 02:57

 ) = 45.4 kJ/mol
) = 45.4 kJ/mol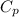 ) = 0.903
) = 0.903 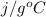
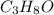 and its mass is 1.12 g. Also, mass of aluminium block is 73.0 g.
and its mass is 1.12 g. Also, mass of aluminium block is 73.0 g.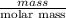
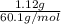
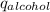 ) = heat lost by aluminium (
) = heat lost by aluminium (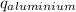 )
)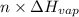 =
= 
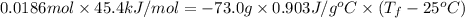
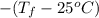
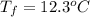
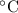 .
.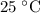
 H)= 45.04 kJ/mol
H)= 45.04 kJ/mol

 45.04 kJ/mol
45.04 kJ/mol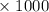 J = - 73
J = - 73 

