
Chemistry, 02.11.2019 07:31 morganhines181
Solution of the schrödinger wave equation for the hydrogen atom results in a set of functions (orbitals) that describe the behavior of the electron. each function is characterized by 3 quantum numbers: n, l, and ml. if the value of n = 1 the quantum number l can have values from to . the total number of orbitals possible at the n = 1 energy level is . if the value of l = 1 the quantum number ml can have values from to . the total number of orbitals possible at the l = 1 sublevel is .

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2


Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
Solution of the schrödinger wave equation for the hydrogen atom results in a set of functions (orbit...
Questions


Mathematics, 20.07.2019 20:40

Physics, 20.07.2019 20:40


Health, 20.07.2019 20:40


Business, 20.07.2019 20:40







Social Studies, 20.07.2019 20:40


Mathematics, 20.07.2019 20:40


Chemistry, 20.07.2019 20:40

Biology, 20.07.2019 20:40

Geography, 20.07.2019 20:40

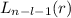 , being n and l the quantum numbers. As is known, in the Laguerre polynomials the subindex must be greater than or equal to 0, which implies
, being n and l the quantum numbers. As is known, in the Laguerre polynomials the subindex must be greater than or equal to 0, which implies 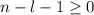 . So if n=1, the only possible value of l is l=0.
. So if n=1, the only possible value of l is l=0. ![Y_{l,m_l}(\theta,\phi)[\tex] as solution. One of the properties of spherical harmonics is that the number [tex]m_l[\tex] can only take the values [tex]m_l=-l,-l+1,-l+2,...,l-1,l[\tex]. So if l=0, as above, [tex]m_l[\tex] can only take the value [tex]m_l=0,[\tex] thus giving only 1 orbital. But if l=1, [tex]m_l](/tpl/images/0356/6953/a304c.png) can be
can be 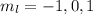 . Therefore the sublevel l=| has 3 possible orbitals.
. Therefore the sublevel l=| has 3 possible orbitals.

