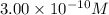Asolution contains 0.021 m cl and 0.017 m i. a solution containing copper (i) ions is added to selectively precipitate one of the ions. at what concentration of copper (i) ion will a precipitate begin to form? what is the identity of the precipitate? ksp(cucl) = 1.0 × 10-6, ksp(cui) = 5.1 × 10-12.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 12:00
Marcel just purchased 1.69 grams of iron fillings in order to make living putty for his 6 year old niece. how many moles of iron are made in his sample?
Answers: 1

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1
You know the right answer?
Asolution contains 0.021 m cl and 0.017 m i. a solution containing copper (i) ions is added to selec...
Questions




Mathematics, 03.02.2020 02:02

Geography, 03.02.2020 02:02


History, 03.02.2020 02:02



Biology, 03.02.2020 02:02

Chemistry, 03.02.2020 02:02



Mathematics, 03.02.2020 02:02

Mathematics, 03.02.2020 02:02


Chemistry, 03.02.2020 02:02

Physics, 03.02.2020 02:02


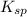 of CuCl =
of CuCl = 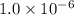
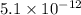
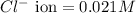
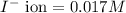
![K_{sp}=[Cu^+][Cl^-]](/tpl/images/0361/6888/73cc7.png)
![1.0\times 10^{-6}=[Cu^+]\times 0.021](/tpl/images/0361/6888/d87ff.png)
![[Cu^+]=\frac{1.0\times 10^{-6}}{0.021}=4.76\times 10^{-5}M](/tpl/images/0361/6888/3549a.png)
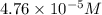
![K_{sp}=[Cu^+][I^-]](/tpl/images/0361/6888/38825.png)
![5.1\times 10^{-12}=[Cu^+]\times 0.017](/tpl/images/0361/6888/99563.png)
![[Cu^+]=\frac{5.1\times 10^{-12}}{0.017}=3.00\times 10^{-10}M](/tpl/images/0361/6888/762ae.png)