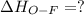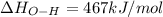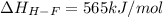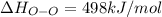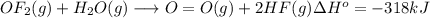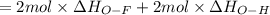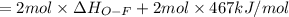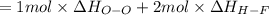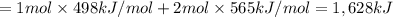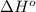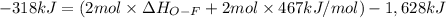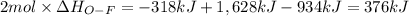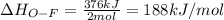Chemistry, 07.11.2019 01:31 jenlicavoli
Oxygen difluoride is an unstable molecule that reacts readily with water. calculate the bond energy of the o–f bond using the standard enthalpy of reaction and the bond energy data provided. just enter a number (no units). of2(g) + h2o(g) \longrightarrow⟶ o=o(g) + 2hf(g) \deltaδh° = –318 kj bond: o–h o=o h–f bond energy (kj/mol): 467 498 565

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 05:00
Asolution is made by dissolving 2.3 moles of sodium chloride (nacl) in 0.155 kilograms of water. if the molal boiling point constant for water (kb) is 0.51 °c/m, what would be the boiling point of this solution? show all the steps taken to solve this problem.
Answers: 1

You know the right answer?
Oxygen difluoride is an unstable molecule that reacts readily with water. calculate the bond energy...
Questions

English, 03.12.2019 02:31


Mathematics, 03.12.2019 02:31

Mathematics, 03.12.2019 02:31



Mathematics, 03.12.2019 02:31

Chemistry, 03.12.2019 02:31



Mathematics, 03.12.2019 02:31




Biology, 03.12.2019 02:31

Mathematics, 03.12.2019 02:31


History, 03.12.2019 02:31

Mathematics, 03.12.2019 02:31