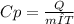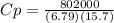Chemistry, 07.11.2019 22:31 jaylan11brown
The heat capacity of a bomb calorimeter was determined by burning 6.79 g of methane (energy of combustion = -802 kj/mol ch4) in the bomb. the temperature changed by 15.7°c. (a) what is the heat capacity of the bomb?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2


Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

You know the right answer?
The heat capacity of a bomb calorimeter was determined by burning 6.79 g of methane (energy of combu...
Questions


Mathematics, 30.11.2020 20:10

English, 30.11.2020 20:10



History, 30.11.2020 20:10

History, 30.11.2020 20:10



Mathematics, 30.11.2020 20:10

Health, 30.11.2020 20:10



Mathematics, 30.11.2020 20:10





Mathematics, 30.11.2020 20:10