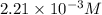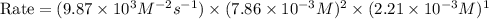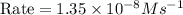Chemistry, 08.11.2019 00:31 giavanleer14
2no + o22 no2 is second order in no and first order in o2. complete the rate law for this reaction in the box below. use the form , where '1' is understood for m, n (don't enter 1) and concentrations taken to the zero power do not appear. rate = in an experiment to determine the rate law, the rate constant was determined to be 9.87×103 m-2s-1. using this value for the rate constant, the rate of the reaction when [no] = 7.86×10-3 m and [o2] = 2.21×10-3 m would be ms-1.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1


Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1
You know the right answer?
2no + o22 no2 is second order in no and first order in o2. complete the rate law for this reaction i...
Questions

Spanish, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10




Mathematics, 09.01.2021 07:10



History, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

English, 09.01.2021 07:10


Mathematics, 09.01.2021 07:10

Biology, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

Mathematics, 09.01.2021 07:10

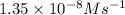
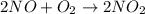
![\text{Rate}=k[NO]^a[O_2]^b](/tpl/images/0364/5919/1903b.png)
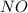 = 2
= 2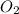 = 1
= 1![\text{Rate}=k[NO]^2[O_2]^1](/tpl/images/0364/5919/3eb46.png)
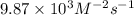
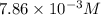
![[O_2]](/tpl/images/0364/5919/b0db0.png) = concentration of
= concentration of