
Chemistry, 12.11.2019 23:31 maskoffvon
How many 3-liter balloons could the 12-l helium tank pressurized to 160 atm fill? keep in mind that an "exhausted" helium tank is not empty. in other words, once the gas inside the tank reaches atmospheric pressure, it will no longer be able to fill balloons. express the number of balloons to three significant figures.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1
You know the right answer?
How many 3-liter balloons could the 12-l helium tank pressurized to 160 atm fill? keep in mind that...
Questions



Mathematics, 06.04.2021 03:40

Health, 06.04.2021 03:40





Mathematics, 06.04.2021 03:40

Mathematics, 06.04.2021 03:40


Mathematics, 06.04.2021 03:40







Mathematics, 06.04.2021 03:40

Mathematics, 06.04.2021 03:40

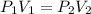
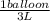 = 636 balloons
= 636 balloons

