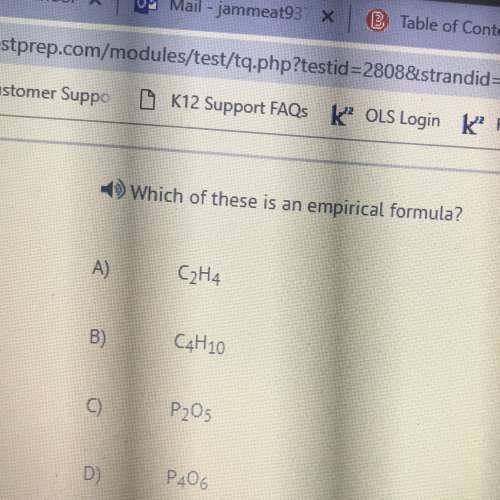
Chemistry, 15.11.2019 19:31 robertss403
Carbon disulfide burns in oxygen to yield carbon dioxide and sulfur dioxide according to the following chemical equation. cs2(l) + 3o2(g)rightwards arrowco2(g) + 2so2(g) b. how many moles of excess reactant remain?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3
You know the right answer?
Carbon disulfide burns in oxygen to yield carbon dioxide and sulfur dioxide according to the followi...
Questions


Mathematics, 16.05.2020 18:57

English, 16.05.2020 18:57

Computers and Technology, 16.05.2020 18:57

History, 16.05.2020 18:57

World Languages, 16.05.2020 18:57

Mathematics, 16.05.2020 18:57

Mathematics, 16.05.2020 18:57


Biology, 16.05.2020 18:57



Mathematics, 16.05.2020 18:57




Social Studies, 16.05.2020 18:57

World Languages, 16.05.2020 18:57

Mathematics, 16.05.2020 18:57

Mathematics, 16.05.2020 18:57