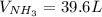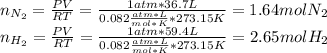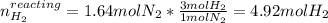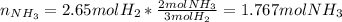Chemistry, 16.11.2019 03:31 kedjenpierrelouis
Consider the reaction between hydrogen gas and nitrogen gas to form ammonia: 3 h2(g) + n2(g) → 2 nh3(g). what volume of ammonia (in l) could be produced by the reaction of 59.4 liters of hydrogen with 36.7 liters of nitrogen at a constant pressure and temperature?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1


Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Consider the reaction between hydrogen gas and nitrogen gas to form ammonia: 3 h2(g) + n2(g) → 2 nh...
Questions


Computers and Technology, 27.07.2021 05:20

Computers and Technology, 27.07.2021 05:20

Advanced Placement (AP), 27.07.2021 05:20

Computers and Technology, 27.07.2021 05:20

Mathematics, 27.07.2021 05:20


Social Studies, 27.07.2021 05:20

Computers and Technology, 27.07.2021 05:20

English, 27.07.2021 05:20


Mathematics, 27.07.2021 05:20








Business, 27.07.2021 05:20