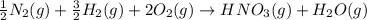Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 3h2(g) 2nh3(g) ah=-92. kj in the second step, ammonia and oxygen react to form nitric acid and water: nh3(9) + 2o2(g) → hno3(9) + h2o(g) ah=-330. kj calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest kj. пkj 1 x ś ?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 15:20
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen...
Questions


Mathematics, 08.11.2019 19:31

English, 08.11.2019 19:31

History, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31



History, 08.11.2019 19:31

English, 08.11.2019 19:31

Social Studies, 08.11.2019 19:31


Mathematics, 08.11.2019 19:31

History, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31

Mathematics, 08.11.2019 19:31


Mathematics, 08.11.2019 19:31



Mathematics, 08.11.2019 19:31

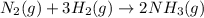 ,
, 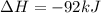
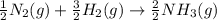 ,
, 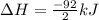
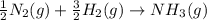 ,
, 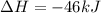 ............ (1)
............ (1)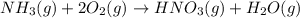
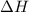 = -330 kJ ............ (2)
= -330 kJ ............ (2)