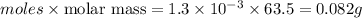Chemistry, 19.11.2019 05:31 nayellisoto15
Acopper cycle experiment takes copper atoms through reactions that produce copper compounds and complexes one after the other, finally producing elemental copper. copper atoms are conserved throughout the process. given that a student begins with 9.29 ml of a 0.14 m cu(no3)2 solution, how much copper should be isolated at the end of the cycle?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Acopper cycle experiment takes copper atoms through reactions that produce copper compounds and comp...
Questions


Social Studies, 19.01.2020 05:31

Social Studies, 19.01.2020 05:31


French, 19.01.2020 05:31



History, 19.01.2020 05:31


History, 19.01.2020 05:31


Chemistry, 19.01.2020 05:31

Geography, 19.01.2020 05:31

Chemistry, 19.01.2020 05:31

Mathematics, 19.01.2020 05:31


English, 19.01.2020 05:31

Advanced Placement (AP), 19.01.2020 05:31


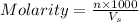
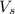 = volume of solution in ml = 9.29 ml
= volume of solution in ml = 9.29 ml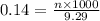
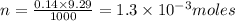
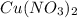 contains 1 mole of copper
contains 1 mole of copper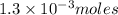 moles of
moles of 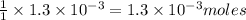 of copper
of copper