Chemistry, 23.11.2019 07:31 langel7373
A23.0 g piece of metal at 99.0 ∘c is placed in a calorimeter containing 53.2 g of water at 24.0 ∘c. the final temperature of the mixture is 26.1 ∘c. what is the specific heat capacity of the metal? assume no energy is lost to the surroundings.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3
You know the right answer?
A23.0 g piece of metal at 99.0 ∘c is placed in a calorimeter containing 53.2 g of water at 24.0 ∘c....
Questions




Computers and Technology, 13.02.2020 23:25



Social Studies, 13.02.2020 23:25


History, 13.02.2020 23:25




Mathematics, 13.02.2020 23:25


English, 13.02.2020 23:25

Mathematics, 13.02.2020 23:25

English, 13.02.2020 23:25

Mathematics, 13.02.2020 23:25

Computers and Technology, 13.02.2020 23:25

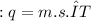
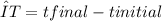 , m the mass and s the specific heat.
, m the mass and s the specific heat.
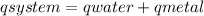
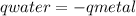
![mw.sw.ΔTw = -[mm.sm.ΔTm ]](/tpl/images/0387/7653/567ae.png)
![53.2 g . 4.184 J/g°C . (26.1 - 24.0)ºC = -[23.0 g . sm . (26.1 - 99.0)°C]](/tpl/images/0387/7653/a0fbc.png)
![464.436 J = -[23.0 g . sm . (-72.9)°C]](/tpl/images/0387/7653/3ce49.png)
![sm = 464.436 J/ -[-1676.7 g°C]](/tpl/images/0387/7653/30209.png)