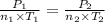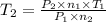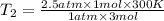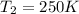Chemistry, 25.11.2019 21:31 ErrorNameTaken505
Aflask of fixed volume contains 1.0 mole of gaseous carbon dioxide and 88 g of solid carbon dioxide. the original pressure and temperature in the flask is 1.0 atm and 300. k. all of the solid carbon dioxide sublimes. the final pressure in the flask is 2.5 atm. what is the final temperature? assume the solid carbon dioxide takes up negligible volume. a. 150 kb. 200 kc. 250 kd. 300 ke. 400 k

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1
You know the right answer?
Aflask of fixed volume contains 1.0 mole of gaseous carbon dioxide and 88 g of solid carbon dioxide....
Questions



Mathematics, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01

Physics, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01


Health, 23.08.2020 02:01


Biology, 23.08.2020 02:01


English, 23.08.2020 02:01

Mathematics, 23.08.2020 02:01

English, 23.08.2020 02:01

English, 23.08.2020 02:01

History, 23.08.2020 02:01

Chemistry, 23.08.2020 02:01


Mathematics, 23.08.2020 02:01