
Chemistry, 27.11.2019 02:31 emilyy4757
Be sure to answer all parts. write a balanced equation and kb expression for the following brønsted-lowry base in water: benzoate ion, c6h5coo−. include the states of all reactants and products in your equation. you do not need to include states in the equilibrium expression. balanced equation: ⇌ kb expression:

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
Be sure to answer all parts. write a balanced equation and kb expression for the following brønsted-...
Questions


Physics, 07.04.2020 22:31


English, 07.04.2020 22:31


Mathematics, 07.04.2020 22:31




Mathematics, 07.04.2020 22:31



Mathematics, 07.04.2020 22:31

History, 07.04.2020 22:31

Mathematics, 07.04.2020 22:31

Mathematics, 07.04.2020 22:31


Mathematics, 07.04.2020 22:31

English, 07.04.2020 22:31

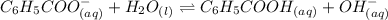
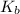 expression is:-
expression is:-![K_{b}=\frac {\left [ C_6H_5COOH \right ]\left [ {OH}^- \right ]}{[C_6H_5COO^-]}](/tpl/images/0392/5825/392ce.png)


