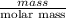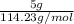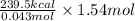Chemistry, 28.11.2019 04:31 gwendallinesikes
During the combustion of 5.00 g of octane, c8h18, 239.5 kcal is released. how much energy is released by the combustion of 1.54 mol of c8h18 ?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
During the combustion of 5.00 g of octane, c8h18, 239.5 kcal is released. how much energy is release...
Questions

History, 26.02.2020 20:53



History, 26.02.2020 20:53










Mathematics, 26.02.2020 20:53

Mathematics, 26.02.2020 20:53


Mathematics, 26.02.2020 20:53



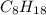 is 114.23 g/mol. Therefore, its number of moles are calculated as follows.
is 114.23 g/mol. Therefore, its number of moles are calculated as follows.