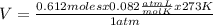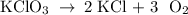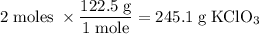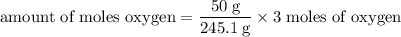Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Where are each of the three particles located within the atom?
Answers: 1


Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2
You know the right answer?
Determine the volume of o2 (at stp) formed when 50.0 g of kclo3 decomposes according to the followin...
Questions



SAT, 28.09.2021 16:30

Social Studies, 28.09.2021 16:30

Social Studies, 28.09.2021 16:30

English, 28.09.2021 16:30

Mathematics, 28.09.2021 16:30


Computers and Technology, 28.09.2021 16:30




History, 28.09.2021 16:30






Mathematics, 28.09.2021 16:40

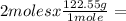 245.1 grams of KClO₃
245.1 grams of KClO₃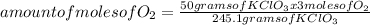
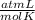 T= 0 C= 273 K
T= 0 C= 273 K