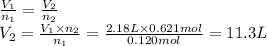Chemistry, 03.12.2019 18:31 leahstubbs
Achemical reaction occurring in a cylinder equipped with a moveable piston produces 0.621 mol of a gaseous product.
if the cylinder contained 0.120 mol of gas before the reaction and had an initial volume of 2.18 l, what was its volume after the reaction?
(assume constant pressure and temperature and that the initial amount of gas completely reacts.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2

Chemistry, 23.06.2019 23:40
What is the difference between a weak and strong base? only some of the molecules of a weak base dissociate to produce hydrogen ions when mixed with water, but all of the molecules of a strong base dissociate to produce hydrogen ions. strong bases result in the production of hydronium, while weak bases produce hydroxide ions. only some of the molecules of a weak base dissociate to produce hydroxide ions when mixed with water, but all of the molecules of a strong base dissociate to produce hydroxide ions. strong bases react with acids to produce water, but weak bases do not react at all.
Answers: 1
You know the right answer?
Achemical reaction occurring in a cylinder equipped with a moveable piston produces 0.621 mol of a g...
Questions

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Chemistry, 15.09.2020 01:01

English, 15.09.2020 01:01

Social Studies, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Geography, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Biology, 15.09.2020 01:01

Chemistry, 15.09.2020 01:01

Geography, 15.09.2020 01:01

Spanish, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

English, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Social Studies, 15.09.2020 01:01

French, 15.09.2020 01:01

Social Studies, 15.09.2020 01:01