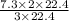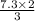Chemistry, 06.12.2019 06:31 KariSupreme
Balance the equation and determine how many liter of nitrogen are required to react with 7.3 l of hydrogen to produce ammonia (nitrogen trihydride)?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1


Chemistry, 23.06.2019 08:00
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3
You know the right answer?
Balance the equation and determine how many liter of nitrogen are required to react with 7.3 l of hy...
Questions


Social Studies, 09.09.2021 22:00



Mathematics, 09.09.2021 22:00

Mathematics, 09.09.2021 22:00











Mathematics, 09.09.2021 22:10

History, 09.09.2021 22:10

Chemistry, 09.09.2021 22:10