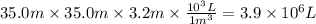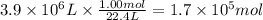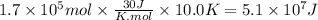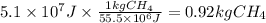Chemistry, 06.12.2019 17:31 keshewar2671
Assuming that all the energy given off in the reaction goes to heating up only the air in the house, determine the mass of methane required to heat the air in a house by 10.0 ∘c. assume each of the following: house dimensions are 35.0 m × 35.0 m × 3.2 m ; specific heat capacity of air is 30 j/k⋅mol; 1.00 mol of air occupies 22.4l for all temperatures concerned.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Llama have 74 chromosomes how many chromosomes will they be found in their gametes explain how you know
Answers: 2

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Assuming that all the energy given off in the reaction goes to heating up only the air in the house,...
Questions

Mathematics, 27.05.2021 08:30

Mathematics, 27.05.2021 08:30



Mathematics, 27.05.2021 08:30


Mathematics, 27.05.2021 08:30

Biology, 27.05.2021 08:30



Biology, 27.05.2021 08:30

Mathematics, 27.05.2021 08:30

Social Studies, 27.05.2021 08:30


Chemistry, 27.05.2021 08:30

History, 27.05.2021 08:30

Advanced Placement (AP), 27.05.2021 08:30



Chemistry, 27.05.2021 08:30