Consider a voltaic cell based on the following cell reaction:
ni(s) + at2(s) ? ni2+(aq) + 2a...

Chemistry, 07.12.2019 02:31 Matildagann
Consider a voltaic cell based on the following cell reaction:
ni(s) + at2(s) ? ni2+(aq) + 2at– (aq)
given that the standard cell emf is 0.55 v, what is the standard reduction potential for astatine? [e°(ni2+/ni) = –0.25 v]

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3

You know the right answer?
Questions

Computers and Technology, 20.01.2022 14:00

Geography, 20.01.2022 14:00

Mathematics, 20.01.2022 14:00

Mathematics, 20.01.2022 14:00

Computers and Technology, 20.01.2022 14:00


Social Studies, 20.01.2022 14:00


English, 20.01.2022 14:00


Mathematics, 20.01.2022 14:00

Mathematics, 20.01.2022 14:00

Mathematics, 20.01.2022 14:00



Mathematics, 20.01.2022 14:00



Mathematics, 20.01.2022 14:00

![E^0_{[At_2/At^-]}=0.30V](/tpl/images/0407/4047/b7e3b.png)
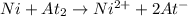
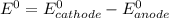
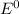 are standard reduction potentials.
are standard reduction potentials.![E^0_{[Ni^{2+}/Mg]}= -0.25V](/tpl/images/0407/4047/bfc73.png)
![E^0_{[At_2/At^-]}=?](/tpl/images/0407/4047/cb54f.png)
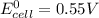
![E^0=E^0_{[At_2/At^-]}- E^0_{[Ni^{2+}/Mg]}](/tpl/images/0407/4047/e55a1.png)
![0.55V=E^0_{[At_2/At^-]-(-0.25V)](/tpl/images/0407/4047/9044a.png)


