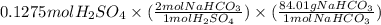Chemistry, 10.12.2019 19:31 Fangflora3
Oh, no! you just spilled 85.00 ml of 1.500 m sulfuric acid on your lab bench and need to clean it up immediately! right next to you there is a small jar labeled "15.00 g sodium bicarbonate". will this be enough sodium bicarbonate to neutralize the spilled sulfuric acid? show your work and state all reasoning!

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Oh, no! you just spilled 85.00 ml of 1.500 m sulfuric acid on your lab bench and need to clean it u...
Questions






Arts, 19.02.2021 02:00




Mathematics, 19.02.2021 02:00


Mathematics, 19.02.2021 02:00

Spanish, 19.02.2021 02:00

Mathematics, 19.02.2021 02:00

Mathematics, 19.02.2021 02:00





Mathematics, 19.02.2021 02:00

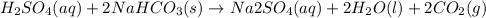
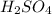 are present in 85.00 mL of 1.500 M sulfuric acid.
are present in 85.00 mL of 1.500 M sulfuric acid.
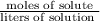
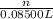
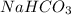 . As, the molar mass of
. As, the molar mass of