
Acertain dose of tums contains 750.0 mg of calcium carbonate, caco3, in each tablet. in this problem, you will follow the steps to calculate how many grams of stomach acid, hcl, are neutralized by one tablet of tums. balance the chemical equation for the reaction of hcl with caco3. chemical equation: hcl + caco_{3} -> cacl_{2} + co_{2} + h_{2}o hcl+caco3⟶cacl2+co2+h2o
using the balanced equation, determine how many moles of hclhcl will react with 1 mol1 mol of caco3.caco3.
moles of hcl: hcl:
calculate how many moles 750.0 mg750.0 mg caco3caco3 is.
750.0 mg caco3=750.0 mg caco3= caco3
using your answers from the first two questions, calculate how many moles of hclhcl are neutralized when 750.0 mg750.0 mg caco3caco3 reacts.
moles of hcl: hcl:
convert the number of moles of hclhcl that you found in the previous question to grams of hcl. hcl. your answer is the amount (in grams) of hclhcl neutralized by one tablet of tums.
mass of hclhcl:

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1
You know the right answer?
Acertain dose of tums contains 750.0 mg of calcium carbonate, caco3, in each tablet. in this problem...
Questions

History, 18.12.2020 19:20

English, 18.12.2020 19:20




Mathematics, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20

Social Studies, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20



English, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20


Mathematics, 18.12.2020 19:20


Mathematics, 18.12.2020 19:20

Mathematics, 18.12.2020 19:20

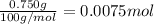
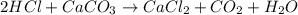
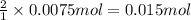 of HCl.
of HCl.

