
Chemistry, 12.12.2019 20:31 stephliu721
A54.3 mg sample of sodium perchlorate contains radioactive chlorine-36 (whose atomic mass is 36.0 amu).
if 29.6% of the chlorine atoms in the sample are chlorine-36 and the remainder is naturally occurring nonradioactive chlorine atoms, how many disintegrations per second are produced by this sample? the half-life of chlorine-36 is 3.0×105 yr.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
You mix the pks of succinic acid are 4.21 and 5.64. how many gramsa graduate student at sdsu wants to measure the activity of a particular enzyme at ph 4.0. to buffer her reaction, she will use a buffer system based on one of the acids listed below, which acid is most appropriate for the experiment? of monosodium succinate (fw = 140 g/mol) and disodium succinate (fw = 162 g/mol) must be added to 1 l of water to produce a solution with a ph 5.28 and a total solute concentration of 100 mm? (assume the total volume remains 1 liter, answer in grams monosodium succinate, grams disodium succinate, respectively.) volumes of 0.05 m nah2po4 and 0.05 m na2hpo4 (pk's for phosphoric acid are 2.15, 6.82 and 12.38). which of the following best describes the resulting solution?
Answers: 2

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3


Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1
You know the right answer?
A54.3 mg sample of sodium perchlorate contains radioactive chlorine-36 (whose atomic mass is 36.0 am...
Questions




Social Studies, 13.06.2021 19:10


Mathematics, 13.06.2021 19:10

History, 13.06.2021 19:10


Mathematics, 13.06.2021 19:10

Engineering, 13.06.2021 19:10


Geography, 13.06.2021 19:10


Mathematics, 13.06.2021 19:10


Mathematics, 13.06.2021 19:10

Biology, 13.06.2021 19:10

Social Studies, 13.06.2021 19:10


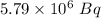
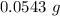
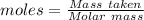
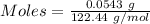
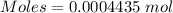
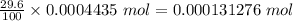
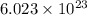 atoms of chlorine
atoms of chlorine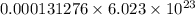 atoms of chlorine
atoms of chlorine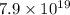
 year
year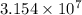 s
s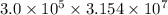 s = 9462000000000 s
s = 9462000000000 s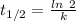
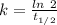
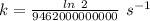
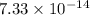 s⁻¹
s⁻¹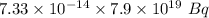 =
= 

