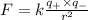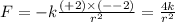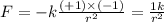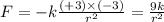Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3


Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
Choose the compound below that should have the highest melting point according to the ionic bonding...
Questions



Arts, 23.10.2020 15:20

Mathematics, 23.10.2020 15:20

English, 23.10.2020 15:20




French, 23.10.2020 15:20

English, 23.10.2020 15:20


Computers and Technology, 23.10.2020 15:20





Advanced Placement (AP), 23.10.2020 15:20


Computers and Technology, 23.10.2020 15:20