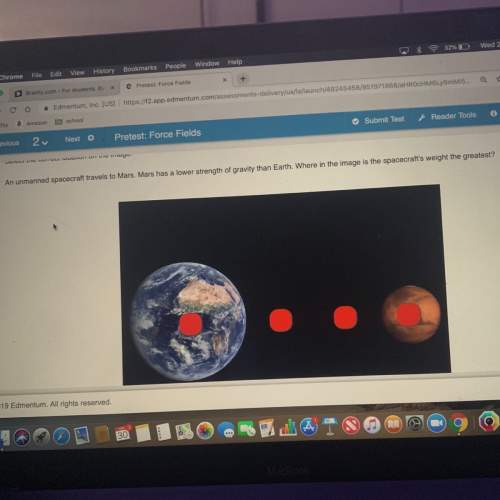
Perform the following for part c of this lab:
a. (4 pts) calculate the volume of naoh that was added to the vinegar. convert this volume to liters. show your work.
b. (2 pts) convert the measured volume of vinegar to liters. show your work.
c. (2 pts) calculate the moles of naoh using the volume and molarity of naoh. show your work. moles = molarity x volume
d. (2 pts) since the reaction ratio is 1: 1, the moles of acetic acid in the vinegar is equal to the moles of naoh reacted during the titration. calculate the molarity of the acetic acid in the vinegar. show your work. molarity = moles / volume
e. (2 pts) calculate the grams of acetic acid in the vinegar. show your work. mass = moles x molar mass (g/mol)
f. (2 pts) assuming that the density of vinegar is very close to 1.0 g/ml, the 2.0 ml sample of vinegar used in the titration should weigh 2.0
g. use this to calculate the mass % of acetic acid in the vinegar sample. mass % = (mass acetic acid / mass vinegar) * 100%

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2
You know the right answer?
Perform the following for part c of this lab:
a. (4 pts) calculate the volume of naoh...
a. (4 pts) calculate the volume of naoh...
Questions

Mathematics, 19.04.2021 14:00


Mathematics, 19.04.2021 14:00


History, 19.04.2021 14:00



Computers and Technology, 19.04.2021 14:00


Mathematics, 19.04.2021 14:00




Mathematics, 19.04.2021 14:00

History, 19.04.2021 14:00




Mathematics, 19.04.2021 14:00

Social Studies, 19.04.2021 14:00