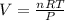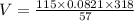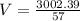Chemistry, 16.12.2019 04:31 alyssaflores
Atank contains 115 miles of neon gas. it has a pressure of 57 atm at a temperature of 45 c. calculate the volume of the tank.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2


Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1
You know the right answer?
Atank contains 115 miles of neon gas. it has a pressure of 57 atm at a temperature of 45 c. calculat...
Questions

Mathematics, 06.01.2021 22:30

Chemistry, 06.01.2021 22:30





Mathematics, 06.01.2021 22:30

Mathematics, 06.01.2021 22:30


Mathematics, 06.01.2021 22:30

Computers and Technology, 06.01.2021 22:40


History, 06.01.2021 22:40


Physics, 06.01.2021 22:40


Business, 06.01.2021 22:40

Mathematics, 06.01.2021 22:40

Mathematics, 06.01.2021 22:40

English, 06.01.2021 22:40