
Alab scientist cools a liquid sample of water (2.6 kg) at 0.00°c to -192°c. the water turns to ice as this temperature change occurs. how much heat is released during this process? [for water, lf = 334 kj/kg and lv = 2257 kj/kg. the specific heat for ice is 2050 j/(kg·k)]. report your answer in kj. (round your answer to a whole number - no decimal places)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
Alab scientist cools a liquid sample of water (2.6 kg) at 0.00°c to -192°c. the water turns to ice a...
Questions








Physics, 11.07.2019 05:20





Health, 11.07.2019 05:20


Geography, 11.07.2019 05:20


History, 11.07.2019 05:20

Biology, 11.07.2019 05:20

Mathematics, 11.07.2019 05:20

Mathematics, 11.07.2019 05:20

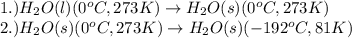
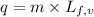 ......(1)
......(1)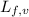 = latent heat of fusion or vaporization
= latent heat of fusion or vaporization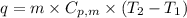 .......(1)
.......(1)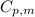 = specific heat capacity of medium
= specific heat capacity of medium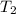 = final temperature
= final temperature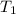 = initial temperature
= initial temperature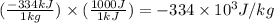
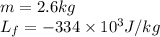
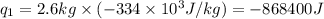

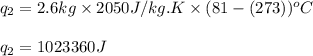

![[-868400+(-1023360)]J=-1891760J=-1891.76kJ\approx -1892kJ](/tpl/images/0420/8409/e23bd.png)


