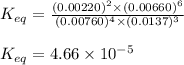9. the first step in industrial nitric acid production is the catalyzed oxidation of ammonia. without a catalyst, a different reaction predominates: 4nh3(g) + 3o2(g) ⇔ 2n2(g) + 6h2o(g) when 0.0120 mol gaseous nh3 and 0.0170 mol gaseous o2 are placed in a 1.00 l container at a certain temperature, the n2 concentration at equilibrium is 2.20×10-3 m. calculate keq for the reaction at this temperature.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1


Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2
You know the right answer?
9. the first step in industrial nitric acid production is the catalyzed oxidation of ammonia. withou...
Questions


Mathematics, 18.10.2020 16:01

English, 18.10.2020 16:01

English, 18.10.2020 16:01

Mathematics, 18.10.2020 16:01





Mathematics, 18.10.2020 16:01


History, 18.10.2020 16:01


Mathematics, 18.10.2020 16:01


Arts, 18.10.2020 16:01

History, 18.10.2020 16:01

Mathematics, 18.10.2020 16:01

Mathematics, 18.10.2020 16:01

English, 18.10.2020 16:01

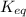 is
is 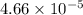
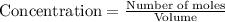
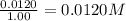
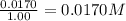
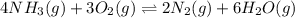
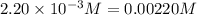
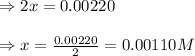
![0.0120-4x=[0.0120-(4\times 0.00110)]=0.00760M](/tpl/images/0421/4577/5e570.png)
![0.0170-3x=[0.0170-(3\times 0.00110)]=0.0137M](/tpl/images/0421/4577/b5ac6.png)
![6x=[6\times 0.00110]=0.00660M](/tpl/images/0421/4577/d0282.png)
![K_{eq}=\frac{[N_2]^2\times [H_2O]^6}{[NH_3]^4\times [O_2]^3}](/tpl/images/0421/4577/7f887.png)