
Hich one of the following statements about orbital hybridization is incorrect?
m
a. the carbon atom in ch4 is sp3 hybridized.
b. the carbon atom in co2 is sp hybridized.
c. the nitrogen atom in nh3 is sp2 hybridized.
d. sp2 hybrid orbitals are coplanar, and at 120 to each other.
e. sp hybrid orbitals lie at 180° to each other.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 22:00
8) warming your hands by a fire is an example if which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 1

Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1

Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3
You know the right answer?
Hich one of the following statements about orbital hybridization is incorrect?
m
a. the...
m
a. the...
Questions



Physics, 22.09.2019 18:50

Biology, 22.09.2019 18:50


Biology, 22.09.2019 18:50


German, 22.09.2019 18:50

Social Studies, 22.09.2019 18:50

Mathematics, 22.09.2019 18:50




Mathematics, 22.09.2019 18:50


Mathematics, 22.09.2019 18:50

History, 22.09.2019 18:50



![\text{Number of electron pair}=\frac{1}{2}[V+N-C+A]](/tpl/images/0422/7584/9e987.png)
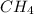
![\text{Number of electrons}=\frac{1}{2}\times [4+4]=4](/tpl/images/0422/7584/b4293.png)
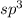 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.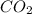
![\text{Number of electrons}=\frac{1}{2}\times [4]=2](/tpl/images/0422/7584/34bd3.png)
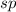 and the electronic geometry of the molecule will be linear.
and the electronic geometry of the molecule will be linear.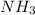
![\text{Number of electrons}=\frac{1}{2}\times [5+3]=4](/tpl/images/0422/7584/5f4d9.png)


