
Chemistry, 19.12.2019 11:31 kaitlyn114433
Need with this next to each formula, write the number of atoms of each element found in one unit of the compound.
these are the compounds below:
1. potassium iodide, ki
2. sodium sulfide, na2s
3. silicon dioxide, sio2
4. carbonic acid, h2co3

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 22.06.2019 15:30
Light waves can move through , but they travel fastest when they move through a(n) .
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
Need with this next to each formula, write the number of atoms of each element found in one unit o...
Questions

Mathematics, 09.01.2020 21:31



Spanish, 09.01.2020 21:31

Chemistry, 09.01.2020 21:31


English, 09.01.2020 21:31


Mathematics, 09.01.2020 21:31





Mathematics, 09.01.2020 21:31


Mathematics, 09.01.2020 21:31

Spanish, 09.01.2020 21:31

Business, 09.01.2020 21:31


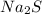 = 3
= 3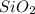 = 3
= 3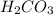 = 6
= 6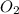 is not an atom , it is molecule of oxygen .
is not an atom , it is molecule of oxygen .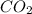 ,
,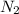 ,
,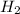 are molecules
are molecules

