
Chemistry, 20.12.2019 04:31 blakesmith0110
Arigid container is divided into two compartments of equal volume by a partition. one compartment contains 1 mole of ideal gas a at 1 atm, and the other contains 1 mole of ideal gas b at 1 atm. calculate the increase in entropy which occurs when the partition between the two compartments is removed. if the frst compartment had contained 2 moles of ideal gas a, what would have been the increase in entropy when the partition was removed? calculate the corresponding increases in entropy in each of the preceding two situations if both compartments had contained ideal gas a.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 12:00
Marcel just purchased 1.69 grams of iron fillings in order to make living putty for his 6 year old niece. how many moles of iron are made in his sample?
Answers: 1
You know the right answer?
Arigid container is divided into two compartments of equal volume by a partition. one compartment co...
Questions






History, 12.08.2020 18:01


History, 12.08.2020 18:01


English, 12.08.2020 18:01



Mathematics, 12.08.2020 18:01

Mathematics, 12.08.2020 18:01

Mathematics, 12.08.2020 18:01

Mathematics, 12.08.2020 18:01

Health, 12.08.2020 18:01

Mathematics, 12.08.2020 18:01

Social Studies, 12.08.2020 18:01

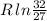 .
.











