Chemistry, 21.12.2019 04:31 MrRandomUser
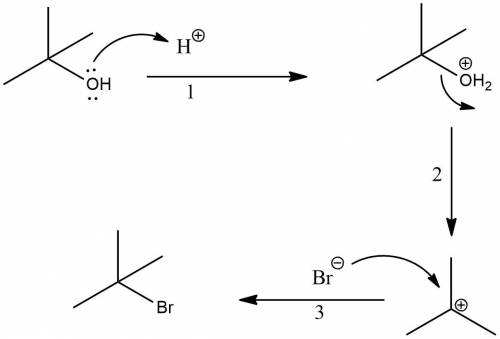
T-butyl bromide (2-bromo-2-methylpropane) can be prepared by simply taking t-butyl alcohol and shaking it with an aqueous solution of hbr at room temperature. the reaction is much faster than with n-butyl alcohol and is essentially 100% complete within a few minutes. give a mechanism for this reaction. what is this type of reaction called

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1

Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
T-butyl bromide (2-bromo-2-methylpropane) can be prepared by simply taking t-butyl alcohol and shaki...
Questions



Spanish, 05.02.2020 10:49



English, 05.02.2020 10:49







History, 05.02.2020 10:50

Mathematics, 05.02.2020 10:50


English, 05.02.2020 10:50




History, 05.02.2020 10:50

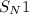 (substitution nucleophilic bimolecular) reaction.
(substitution nucleophilic bimolecular) reaction.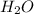 .
.