
Chemistry, 07.01.2020 08:31 angelblalock5885
Which intermolecular forces do the particles in the image (f– in water) experience? note that the ion is not necessarily drawn to scale.
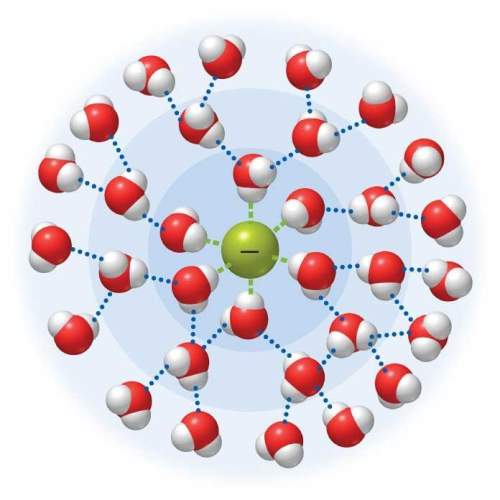

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 11:00
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Which intermolecular forces do the particles in the image (f– in water) experience? note that the i...
Questions


Social Studies, 08.03.2021 18:40


English, 08.03.2021 18:40


Biology, 08.03.2021 18:40

Mathematics, 08.03.2021 18:40

Advanced Placement (AP), 08.03.2021 18:40

Mathematics, 08.03.2021 18:40

English, 08.03.2021 18:40


Mathematics, 08.03.2021 18:40


Mathematics, 08.03.2021 18:40


English, 08.03.2021 18:40

Mathematics, 08.03.2021 18:40


Mathematics, 08.03.2021 18:40

English, 08.03.2021 18:40



