Chemistry, 09.01.2020 06:31 aangellexith2885
Suppose you add 100 grams of water at 60.0 degrees c to 100
gramsice at 0.00 degrees c. some of the ice melts and cools thewarm
water to 0.00 degrees c. when the ice/water mixture hascome to a
uniform temperature of 0.00 degrees c, how much ice
hasmelted?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 00:30
Fred is studying a substance that is made out of only one element. this means that
Answers: 1
You know the right answer?
Suppose you add 100 grams of water at 60.0 degrees c to 100
gramsice at 0.00 degrees c. some o...
gramsice at 0.00 degrees c. some o...
Questions



Mathematics, 16.04.2020 20:32


Social Studies, 16.04.2020 20:32


Mathematics, 16.04.2020 20:32


History, 16.04.2020 20:32

Computers and Technology, 16.04.2020 20:32




Mathematics, 16.04.2020 20:32

Law, 16.04.2020 20:32

Physics, 16.04.2020 20:32

Mathematics, 16.04.2020 20:32

Mathematics, 16.04.2020 20:32

History, 16.04.2020 20:32

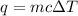
 = change in temperature =
= change in temperature = 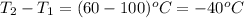
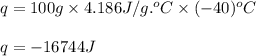
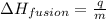
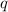 = amount of heat absorbed = 16744 J
= amount of heat absorbed = 16744 J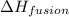 = heat of fusion = 333 J/g
= heat of fusion = 333 J/g