

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1
You know the right answer?
How much potassium chlorate is needed to produce 20.0 ml
ofoxygen gas at 670 mm hg and 293 k?...
ofoxygen gas at 670 mm hg and 293 k?...
Questions


Law, 15.10.2021 01:40

Mathematics, 15.10.2021 01:40

Business, 15.10.2021 01:40


Mathematics, 15.10.2021 01:40

Mathematics, 15.10.2021 01:40

Mathematics, 15.10.2021 01:40


Mathematics, 15.10.2021 01:40






Medicine, 15.10.2021 01:40

Social Studies, 15.10.2021 01:40



Chemistry, 15.10.2021 01:40

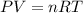
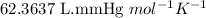

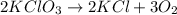
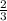 moles of potassium chlorate reacts
moles of potassium chlorate reacts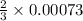 moles of potassium chlorate reacts
moles of potassium chlorate reacts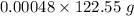 = 0.058824 g
= 0.058824 g

