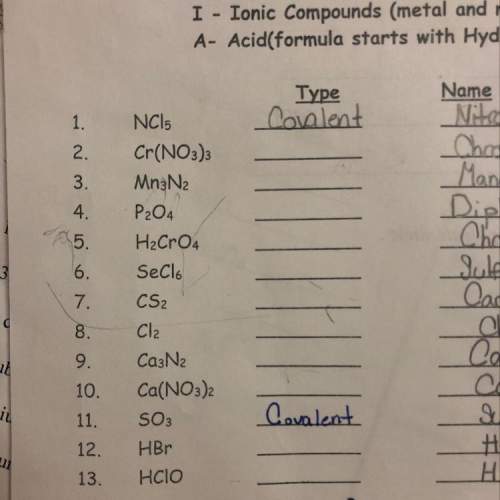
Which ones are covalent, and ionic?
...

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1
You know the right answer?
Questions

Mathematics, 24.04.2021 01:00


Arts, 24.04.2021 01:00

World Languages, 24.04.2021 01:00

English, 24.04.2021 01:00

Mathematics, 24.04.2021 01:00

Mathematics, 24.04.2021 01:00

Mathematics, 24.04.2021 01:00

Biology, 24.04.2021 01:00




Mathematics, 24.04.2021 01:00

Chemistry, 24.04.2021 01:00


Mathematics, 24.04.2021 01:00

Mathematics, 24.04.2021 01:00



Mathematics, 24.04.2021 01:00