
Chemistry, 20.01.2020 21:31 brisacruz013
Asolution of chloroform, chcl3, and acetone, (ch3)2co, exhibits a negative deviation from raoult's law. this result implies that

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Where are each of the three particles located within the atom?
Answers: 1

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
Asolution of chloroform, chcl3, and acetone, (ch3)2co, exhibits a negative deviation from raoult's l...
Questions

English, 27.09.2020 14:01


Mathematics, 27.09.2020 14:01


Biology, 27.09.2020 14:01

Mathematics, 27.09.2020 14:01

Chemistry, 27.09.2020 14:01


Mathematics, 27.09.2020 14:01


Chemistry, 27.09.2020 14:01

English, 27.09.2020 14:01



Mathematics, 27.09.2020 14:01

Arts, 27.09.2020 14:01

Mathematics, 27.09.2020 14:01


English, 27.09.2020 14:01

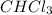 ) and acetone (
) and acetone (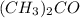 ) exhibits a negative deviation from Raoult's law. This result implies that:
) exhibits a negative deviation from Raoult's law. This result implies that:


