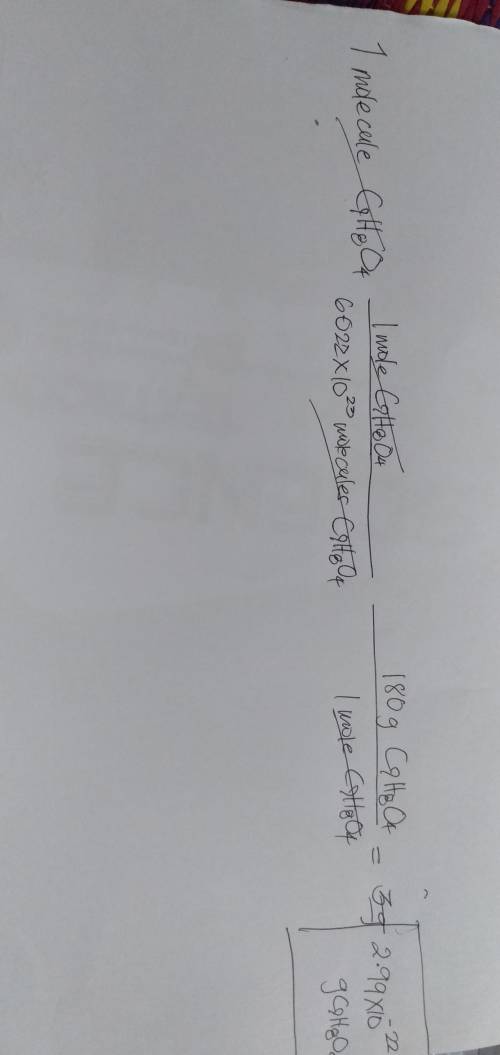
Chemistry, 22.01.2020 07:31 asiamuhammad6
What is the mass, in grams, of a molecule of aspirin (c9h8o4)?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2


You know the right answer?
What is the mass, in grams, of a molecule of aspirin (c9h8o4)?...
Questions

Social Studies, 06.11.2020 07:40

Mathematics, 06.11.2020 07:40


Mathematics, 06.11.2020 07:40

Biology, 06.11.2020 07:40



English, 06.11.2020 07:40

Mathematics, 06.11.2020 07:40



Mathematics, 06.11.2020 07:40

French, 06.11.2020 07:40

History, 06.11.2020 07:40

History, 06.11.2020 07:40

English, 06.11.2020 07:40

Social Studies, 06.11.2020 07:40


Mathematics, 06.11.2020 07:40