The compound that has been least ionic is 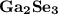 . The correct option is C.
. The correct option is C.
Ionic compounds have been formed with the electrostatic interaction between the oppositely charged ions. The ionic bond has been formed by the transfer of electrons.
The ionic strength of the compound can be assessed by the difference in the electronegativity of the atoms involved in the bond.
The difference in electronegativity of the following compounds has been as follows:
A. NaCl
Difference = 3 - 0.9
Difference = 2.1
B. NaF
Difference = 4- 0.9
Difference = 3.1
C. 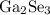
Difference = 2.4 - 1.6
Difference = 0.8
D. ZnO
Difference = 3.5 - 1.6
Difference = 1.9
E. 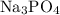
Difference = 3.5 - 0.9
Difference = 2.6
The ionic compound with the least difference in electronegativity has been termed to be the least ionic in nature.
The smallest electronegativity difference has been observed in 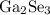 . Thus the compound that has been least ionic is
. Thus the compound that has been least ionic is 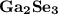 . The correct option is C.
. The correct option is C.
For more information about the ionic strength of the compound, refer to the link:
link
























