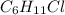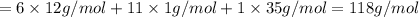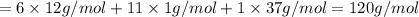Halogenated compounds are particularly easy to identify by their mass spectra because chlorine and bromine occur naturally as mixtures of two abundant isotopes. chlorine occurs as 35cl (75.8%) and 37cl (24.2%); bromine occurs as 79br (50.7%) and 81br (49.3%). for the compound chlorocyclohexane, c6h11cl: at what masses do the molecular ions occur? (list in order of increasing mass separated by commas, e. g. 120,122.) what are the percentages of each molecular ion?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1


Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3
You know the right answer?
Halogenated compounds are particularly easy to identify by their mass spectra because chlorine and b...
Questions







Mathematics, 02.08.2019 10:30

Mathematics, 02.08.2019 10:30




Social Studies, 02.08.2019 10:30

Geography, 02.08.2019 10:30




Biology, 02.08.2019 10:30